Four Key Device Trends Driving the Medical Connector Market
Trends driving collaboration between medical device and connector manufacturers include increased demand for safety and regulatory compliance, disposable solutions, high-resolution imaging capabilities, and durability.
The quality and reliability of electronic equipment for the medical industry is vital, as human life and safety depend on it. This formidable responsibility not only impacts the device manufacturer, but the suppliers of every enabling component within that equipment.
Doctor operation in operation room at hospital concept for insurance advertising.Pediatric surgeon at work.hospital brochures.
To help optimize patient outcomes, medical device manufacturers develop innovative devices and update existing devices with new electronic features and capabilities. These manufacturers rely heavily on their connector manufacturer partners to deliver solutions that will effectively drive their most innovative product designs while also meeting size, cost, performance, and compliance requirements. To support their manufacturing partners, connector suppliers often draw upon their experience in other areas of the diverse medical device industry, as well as in other connector-critical market sectors — including the aviation, aerospace, and automotive industries — to overcome challenges and enable the next generation of medical devices. Current medical market trends driving collaboration between medical device and connector manufacturers include increased demand for safety and regulatory compliance, lower-cost disposable solutions, higher-resolution imaging capabilities, and enhanced durability.
Improved safety and regulatory compliance
As medical devices continue to integrate additional features and functional capabilities, they grow more susceptible to effects of electrostatic discharge (ESD). Although static shocks are a widespread phenomenon, they can be hazardous in medical environments — causing software to freeze, reboot, or malfunction, damaging delicate circuity, or even shocking patients and operators. As a result, the International Electrotechnical Commission (IEC) nearly doubled its ESD performance requirements in the fourth edition of the IEC 60601-1-2:2014 standard, in effect since December 31, 2018. The minimum voltage that a panel-mounted receptacle must withstand is now 15kV, measured from the receptacle housing to the internal electrical contacts, which represents a dramatic increase from the 8kV minimum mandated by the previous edition of the standard.
To help medical device manufacturers comply with this potentially challenging regulation, LEMO took a two-pronged approach. To address project developments with challenging connectivity demands, plastic REDEL SP Series connectors offer advanced features including eight additional high-density electrical contacts in the same small form factor and a tested ESD resistance of 25kV, which not only meets the IEC standard but provides medical device designers plenty of room for future-proofing ensuing designs. To address current applications that required backward compatibility, LEMO subjected its popular REDEL 1P Series plastic connectors to additional testing. They achieved an ESD rating of 13kV right out of the gate, which was well over the incumbent standard, but still slightly short of the new one. The engineering team’s elegant solution consisted of simply adding a thicker dress nut to these highly dependable and widely employed connectors. This allowed customers could meet the demands of the new standard without a costly redesign.

LEMO REDEL SP self-latching, plastic, push-pull connectors have an ergonomic grip, color-coding and keying options, high contact density, and an IP50 seal. They also exhibit high resistance to chemicals, shock, and sterilization processes and are 100% scoop-proof and compatible with Northwire BioCompatic silicone cable alternative.
The new IEC 60601 ESD standard is also providing an impetus for device manufacturers to reconsider longstanding equipment design aspects and proactively build in new capabilities, above and beyond those currently required by the new ESD standard. For example, some manufacturers are further improving the safety, regulatory compliance compatibility, and functional capability of device designs by specifying touch-proof hybrid connectors with higher pin counts that offer additional benefits, including better sensors and imaging capabilities, and surpass the new 15kV ESD requirement. Touch-proof connectors enshroud all the metal components within a plastic housing so there is no chance of inadvertent contact with patients, doctors, or other personnel and can also incorporate 360° internal metallic shielding to protect devices against electromagnetic and radio-frequency interference (EMI) in noisy environments. This can be a valuable upgrade in medical applications with high signal integrity requirements.
Lower-cost disposable solutions
Coupled with the longstanding desire for lower-cost medical solutions is an increasing trend toward disposable devices. The significant cost constraints levied against disposable components present a real engineering challenge, especially since these devices require advanced performance capabilities even though they will only be used for one procedure and then thrown away. For example, the demand for higher-resolution imaging and mapping capabilities requires higher-density electrical contacts, but imaging equipment is one of the many medical device segments increasingly employing disposable devices. So, connector manufacturers must come up with creative solutions for minimizing cost while simultaneously enhancing capabilities.
Minimizing cost is an even greater concern in disposable medical applications than in reusable devices that can be readily sterilized many times. From the connector manufacturers’ point of view, the properties that can’t be compromised — like operating performance and reliability — must remain unchanged, while all those that can — like longevity — must be analyzed for potential cost-saving opportunities. Strategies include using high-quality but lower-cost materials that might be less mechanically or chemically robust than those employed in connectors designed to withstand thousands of steam cycles and mitigating the use of any high-cost materials still required by, for instance, reducing gold-plating thicknesses. Simplifying manufacturing procedures is another tactic and can include designing products in fewer parts, thereby utilizing fewer molds and simplifying assembly, or making greater use of automated assembly processes.
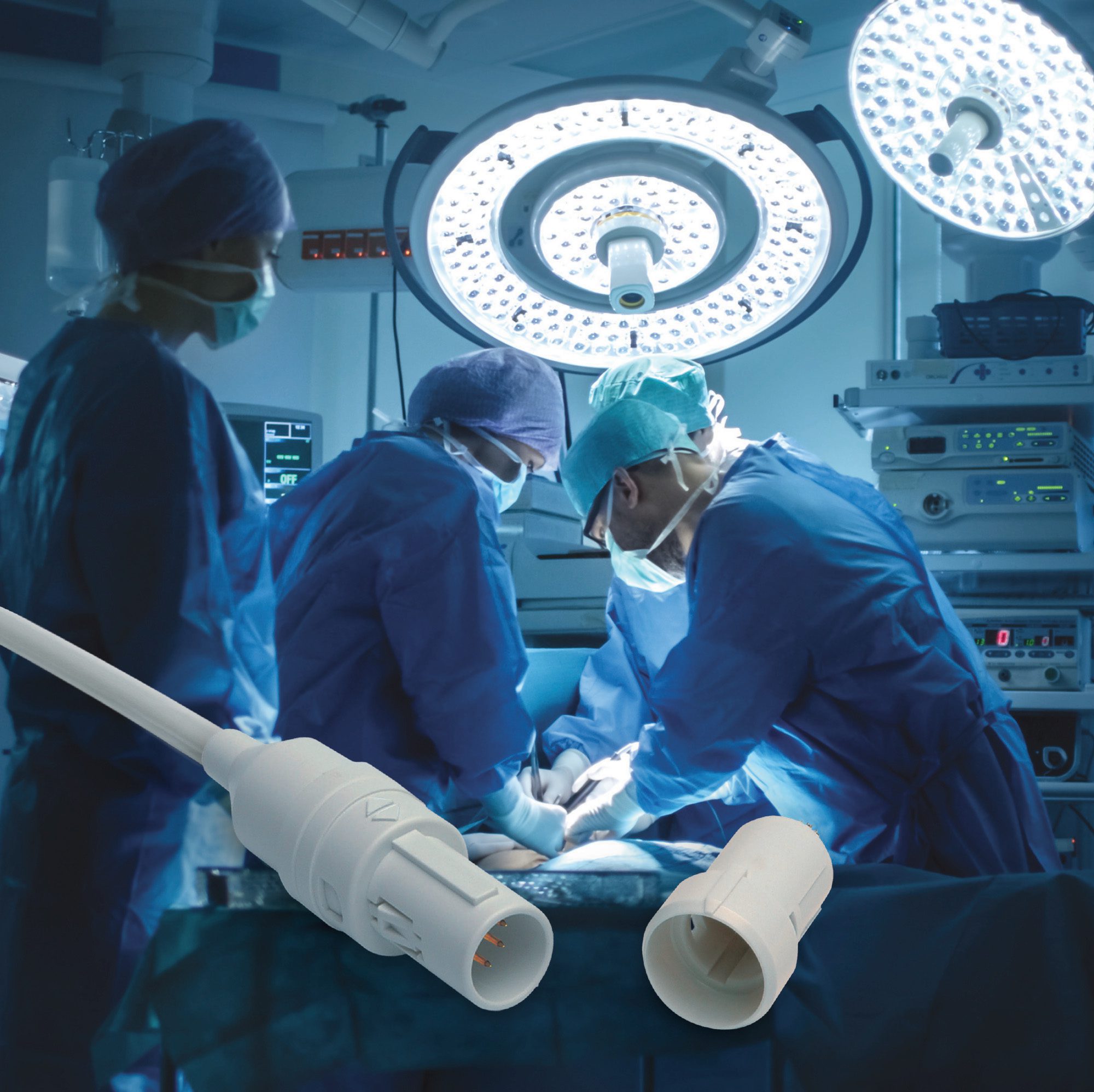
LEMO REDEL Single-Patient-Use Connectors for disposable medical applications minimize material to economically justify a single use while retaining critical quality attributes of the reusable, sterilizable REDEL medical connectors they’re made to mate with, such as high-reliability gold-plated contacts.
In addition to driving a portion of the demand for disposable devices, the widespread demand for higher-resolution medical imaging capabilities is leading many medical device manufacturers to incorporate fiber optic connectivity technologies. Fiber optic technology is highly effective for transmitting high-speed, high-bandwidth data, like high-frequency imaging signals. The challenge lies in training end-users to clean the fibers regularly and properly ensure optimal performance.
Enhanced durability
Manufacturers are continually looking for better ways to protect the integrity of higher-end devices against the potential ravages of medical hazards, including multiple autoclave sterilization cycles. One of the most effective approaches is to enhance the durability of the device and its enabling components. To this end, connector manufacturers can utilize welded glass-to-metal or ceramic-to-metal hermetic vacuum seals, which provide a superior degree of sealing integrity and are used in other high-reliability industries, including aerospace and extractive energy industries. The glass used in these applications is processed and fused at high temperatures around the metal, which effectively makes it break-resistant and prevents the leakage or ingress of liquid or gas of any kind. Hermetic seals are tested based on how many helium atoms can pass through at a certain leakage rate and are thus vastly superior to O-ring, epoxy, and other traditional sealing technologies. This can also be accomplished using a broad range of different glass, metal, and/or ceramic materials to achieve an array of different combinations with various mechanical and insulating properties and costs.