Cutting the Cord: Wireless Charging Technology for Medical Devices
Developing new wireless medical technologies involves more than great design. Learn about the Qi standard, charging strategies, and other steps in the process between concept and completed product.

Advanced wireless charging technologies are transforming the market for portable medical devices, with new equipment for health monitoring, therapeutics, diagnostics, fitness, and wellness changing the healthcare landscape. The advent of lower-cost sensors, energy-efficient devices, high-speed wireless connectivity, and improved charging technologies have enabled a projected market growth from $15 billion in 2020 to $33 billion by 2031, a CAGR of 11%. New technologies enable patients to care for their health at home with respiratory monitoring and assistance devices such as CPAP machines, apnea monitors, pulse oximeters, cardiac monitors, electrocardiogram devices, blood glucose monitors, insulin infusion pumps, and ultrasound devices.
Wireless medical devices offer higher mobility, reliability, convenience, and ease of use for medical personnel and patients. Wireless charging systems also eliminate the need for disposable batteries and charging cords, reducing their environmental impact. Eliminating external charging cords also makes it easier to design watertight medical device enclosures for easy sterilization or explosion-proof devices for specialized healthcare environments.

Wireless charging circuitry for medical electronic devices.
Wireless charging for portable devices
Designing the next generation of medical technologies is a collaborative process involving medical experts, design professionals, components suppliers, and standards organizations. In such efforts, BizLink has successfully developed extensive capabilities for the design and manufacture of wireless charging systems for medical devices, automotive systems, and other applications.
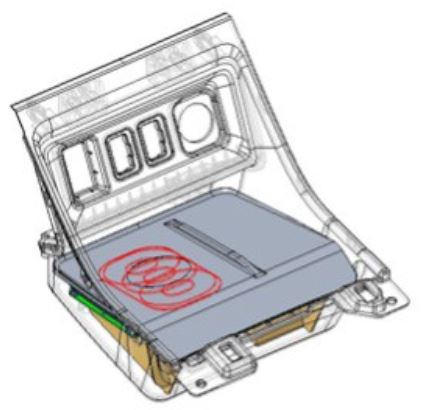
Wireless charging system for vehicle applications.
In one example, BizLink helped a medical device OEM develop a wireless charging system for a family of innovative portable pulsed-wave Doppler ultrasound scanners. The handheld devices feature linear, curved, or phased array ultrasound signals that are transmitted via Wi-Fi to a smartphone to provide medical professionals with the ability to do full-body scans with crisp, full-color images and video. The wireless charging system for the ultrasound scanners allows them to be easily disinfected. BizLink engineers reviewed the customer’s requirements and designed all aspects of the product, including mechanical design, electronics, packaging, accessories, and even the user manual for the product, produced in more than 40 languages. BizLink also conducted a series of more than 20 tests for compatibility, reliability, compliance, ingress protection, atmospheric (air pressure), and foreign object detection (FOD). Using its network of local representatives for certification applications in more than 250 countries.
Global standards guide wireless medical device design
As wireless charging continues to evolve beyond smartphones and consumer devices into medical monitors and diagnostic equipment, standards ensure safety, performance, and security for these applications. A group of more than 350 member companies formed the Wireless Power Consortium (WPC) to establish an open collaborative set of wireless charging standards.
The leading WPC standard used in portable medical devices is Qi (pronounced “chee”), designed for power sources ranging from 5 to 30 watts (classified as Power Class 0). Qi Certification requires test procedures for safety, interoperability, and usability, conducted by a worldwide network of independent authorized test labs. The testing ensures that all devices bearing the Qi logo are compatible with each other, regardless of manufacturer, country of origin, and other factors.
Qi certification testing involves submitting the product to an authorized test lab (ATL) for safety, performance, and compatibility tests. It must also pass interoperability testing at an interoperability testing center (IOC) to ensure compatibility with other Qi devices and systems.

Accessory cable for portable medical electronic device.
One-stop design-to-manufacturing capabilities simplify the development of wireless medical devices
The collaborative design process begins with a thorough analysis of customer requirements for their medical device, including the mechanical design of the enclosure, all electronic circuitry, product packaging at all levels (including labeling, boxes, cartons, and palletization), as well as accessories such as cabling and the development of the user manual. In evaluating customer designs, BizLink will often recommend enhancements to the design for increased performance or reduced cost.
Once the design is established, the BizLink engineering team conducts an extensive multi-level analysis before the product goes to manufacturing, including electronic circuit testing and optimization, electromagnetic compliance, thermal management, and mechanical strength. BizLink’s manufacturing facility is certified to internationally agreed ISO 13485 standards, which sets out requirements for a quality management system and ensure best practices specific to the medical devices industry. The BizLink medical facility is designed for high-mix, low-volume operations and can support a wide range of medical device manufacturing.
Medical device manufacturers can benefit from an experienced partner who can provide not only manufacturing services for their wireless charging systems, but also design optimization, certification testing, and packaging services to deliver a complete turnkey solution for safe and effective wireless charging systems.
For more information on high-performance wireless charging solutions, visit BizLink online.
Like this article? Check out our other miniaturization and standards articles, our Medical Market Page, and our 2023 and 2022 Article Archive.
Subscribe to our weekly e-newsletters, follow us on LinkedIn, Twitter, and Facebook, and check out our eBook archives for more applicable, expert-informed connectivity content.