Medical Connectors: Measuring Creepage and Clearance for Performance and Compliance
Electrical connectors play a key role in advancing product capabilities and ensuring consistent, reliable performance in medical electrical and electronic equipment, especially in today’s IT- enabled and interoperable world.
By Patrick Kinyanjui, Principal Engineer, Fischer Connectors, Inc.
Connectors are an important element to consider when developing a risk assessment for a new or updated device. When consistent, reliable performance and risk management are on the line, understanding the specifications for creepage and clearance is critical.
Managing risk in medical device manufacturing is shaped by international regulations that impact device design right down to the component level. The International Electrotechnical Commission’s 60601-1 (IEC 60601) provides a widely accepted benchmark for medical electronic equipment, and many companies view compliance with this regulation as the de facto requirement for most markets. This standard requires companies to assess the safety and risk factors that the medical devices they design and manufacture pose to patients, health care workers, and maintenance technicians. In the US, the FDA also provides strict regulations to ensure safety. To manage risk and ensure compliance with these rules, many manufacturers have adopted formal risk management methodologies.
Measuring Creepage Distance and Air Clearance
One example of the standard’s technical requirements for connectors is the standard definitions of and new requirements for creepage distance and air clearance, both of which are critical for identifying how a device reduces risk to patients and operators. Device manufacturers must consider how a connectors’ creepage distance and air clearance increases or decreases the risks associated with their devices.
To make the correct assessment, it’s important to understand how creepage and clearance are defined. Creepage is the shortest distance on the surface of an insulating material between two conductive elements, while clearance is the shortest distance through the air between two conductive elements. (See Figure 1.)
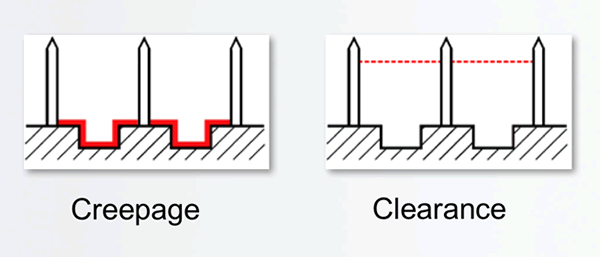
Figure 1: Measuring creepage vs. measuring clearance.
Measuring creepage and clearance can be tricky, but developers need to understand how to properly measure these factors in order to comply with risk management requirements. When a gap of any depth between two conductive elements is less than 1mm wide, both creepage distance and air clearance are measured point-to-point, as if the gap didn’t exist. However, when the gap between two conductive elements is greater than or equal to 1mm, creepage distance must be measured along the contour of the groove, on the surface of an insulating material between two conductive elements, even though air clearance is still measured point-to-point across the gap. (See Table 1, Examples 1 and 2.) The exception to these conditional rules applies to uncemented joints, which also require line-of-sight measurements for both creepage distance and air clearance. (See Table 1, Example 3.)
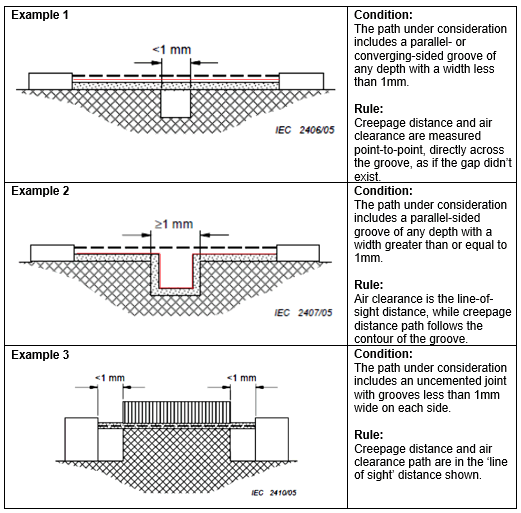
Table 1: Conditional rules for measuring creepage distance and air clearance.
Understanding the rules regarding creepage distance and air clearance helps medical device manufacturers specify connectors that comply with technical requirements, satisfy the essential performance characteristics required for their devices, and eliminate unacceptable risks.
Additional Connector and Cable Considerations for Performance and Compliance
Other important technical considerations when specifying connectors and cabling for the manufacture of medical electrical equipment and systems include ease of implementation, minimizing physical size while maximizing capabilities (e.g., combining signal and power in a single connector), aesthetic appeal, sterilization capabilities and limitations, EMI/RFI shielding, and cost-effectiveness. Device engineers should be able to provide detailed guidance about these and other relevant application requirements so component engineers can provide them with connector and cabling options that are suitable for the medical device application at hand.
Medical equipment developers also need to understand the rules defined in IEC 60601 and work closely with their connector and cable suppliers to make sure they are designing equipment not only for function, but for compliance as well. Defining the essential performance requirements and related technical specifications for connectors and cables is critical to ensuring that the risk assessment profile is accurate, and an accurate risk assessment profile is critical to ensuring both regulatory compliance and desired performance.
Checklist for Specifying Medical Connectors
To help simplify connector and cabling design and selection, medical equipment manufacturers should consider the items in the following checklist.
- Number of connectors needed
- Voltage and current ratings of each contact
- Known creepage distance and air clearance requirements for each contact
- Environmental and sterilization requirements
- Material requirements and use (e.g., metal vs. plastic, reusable vs. disposable)
- Tests to be performed on the completed assembly
- Cable requirements for signal or power (e.g., twisted pairs, coax, etc.)
Cabling requirements may also impact the interconnect system, and should be carefully considered alongside connector requirements in order to accurately determine the risk of any system, but especially those destined for employment in medical devices.





